Identification of foreign particulate matter in assembled autoinjectors
Abstract
The integrity of parenteral products is critical to patient safety, requiring these products to be free from visible particles that may indicate sterility issues. WILCO AG introduces an advanced Automated Visual Inspection (AVI) method for assembled autoinjectors, leveraging long-time tracking of individual object trajectories to distinguish between foreign particulate matter and harmless gas bubbles. This white paper outlines the innovative approach, the design and engineering processes, and the benefits of WILCO AG’s technology, setting a new standard for quality assurance in pharmaceutical manufacturing. Our method significantly enhances the detection rate of foreign particles while minimizing false rejects as well as production costs.
Download White Paper
1. Introduction
Parenteral products are subject to rigorous quality control standards, necessitating the absence of visible particles to prevent adverse health effects, as established by the United States Pharmacopoeia Chapter on visible Particles in Injections (USP <790> Visible Particulates in Injections).
Conventional inspection methods are challenged by the variety of physical properties of these particles (e.g. size, color, shape, density and reflectivity), of the solutions they inhabit (such as color, viscosity, opacity, and surface tension) and of the primary packaging (material, color, resistance to stress). This complexity explains the probabilistic nature of the visual inspection process. Consequently, false rejects as well as false accept cannot be completely ruled out, potentially undermining production efficiency and product safety.
WILCO AG’s method revolutionizes the detection and identification of foreign particles in liquid solutions within assembled autoinjectors, ensuring high product integrity.
2. WILCO's unique approach
WILCO AG has developed a unique methodology for the precise identification and localization of foreign particles within assembled autoinjectors through long-time tracking of individual objects’ trajectories. This approach is based on the specific behavior of particles in liquid solutions - whether they float, sink, or swim. Tailored systems for handling and transportation facilitate the effective mobilization and detection of these particles.
3. Solution Design Process as Key of Success
When designing tailor-made Container Closure Integrity Testing and Automated Visual Inspection systems, we always collaborate closely with our customers – no matter how well defined their needs are. Our aim is to follow a structured and risk-based approach to build a perfectly tailored system for our customer’s products to meet the complex, regulatory requirements.
To guide this co-creation, we have developed our holistic Solution Design Process with 5 phases.
3.1 Feasibility Study
At the beginning, the existing production processes, the requirements, the packaging, the product and the defects are analyzed together with the customer in a holistic approach. This was followed by a risk analysis (FMEA).
The main goal of the Feasibility Study is to design the optical setups to perform the inspection tasks defined together with our customers during the Knowledge Gathering and based on the results of the Risk Assessment.
Our unique approach consists in using a static optical path (OptiXTM), where the optical elements are fixed, thus avoiding misalignments and wear out of optical elements. For the identification and localization of foreign particulates, a test system consisting of an optical setup in the backlight configuration and a robotic arm that turns the autoinjector upside-down mimicking the handling by the manual inspector has been built. The integration of a robotic arm enables on one side to test several translation and rotation profiles, thanks to the 6-axis movement, and on the other side to achieve a great reproducibility in the handling and presentation of the autoinjector in front of the optical setup.

4. Transport and Parallelization
The developments of the pre-engineering phase, that involves several steps to ensure thorough particle mobilization and inspection of the liquid solution, are translated into the final machine concept with a linear transport system integrating handling and movement of the autoinjectors in front of the optical system:
- Transport of the autoinjectors to the test system for inspection via a linear conveyor.
- Upside-down inversion of the autoinjectors before image acquisition to mobilize particles eventually adherent to internal surfaces and release them back into the liquid solution.
- Orientation adjustments of adjacent autoinjectors to prevent collisions.
- Parallelization of test systems to inspect 12 autoinjectors simultaneously at a rate of 300 Autoinjectors per minute.
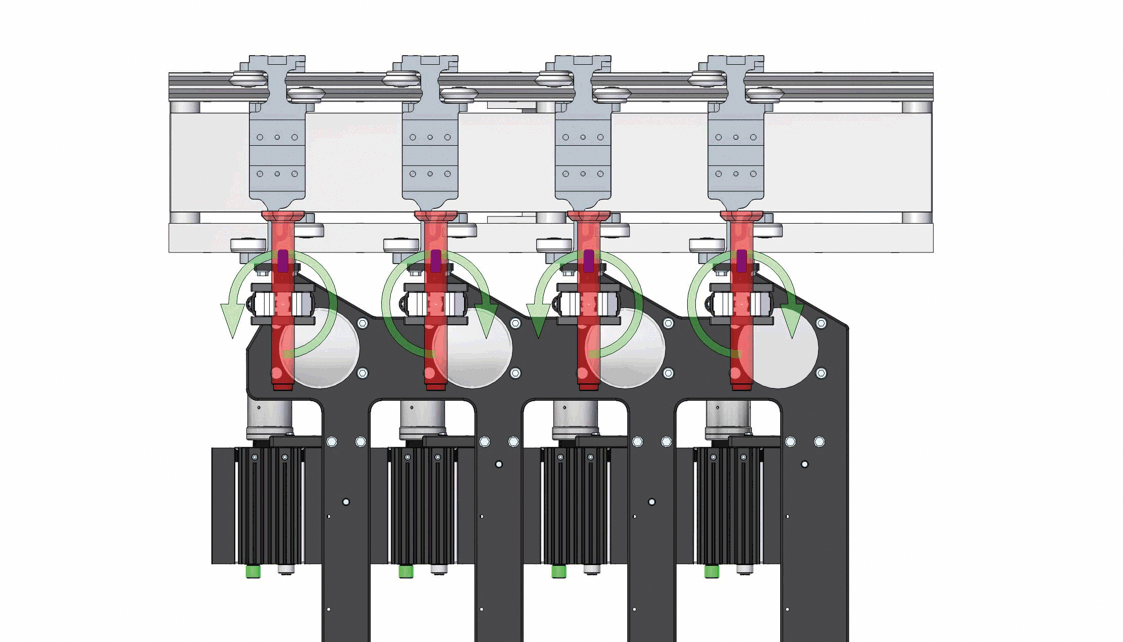
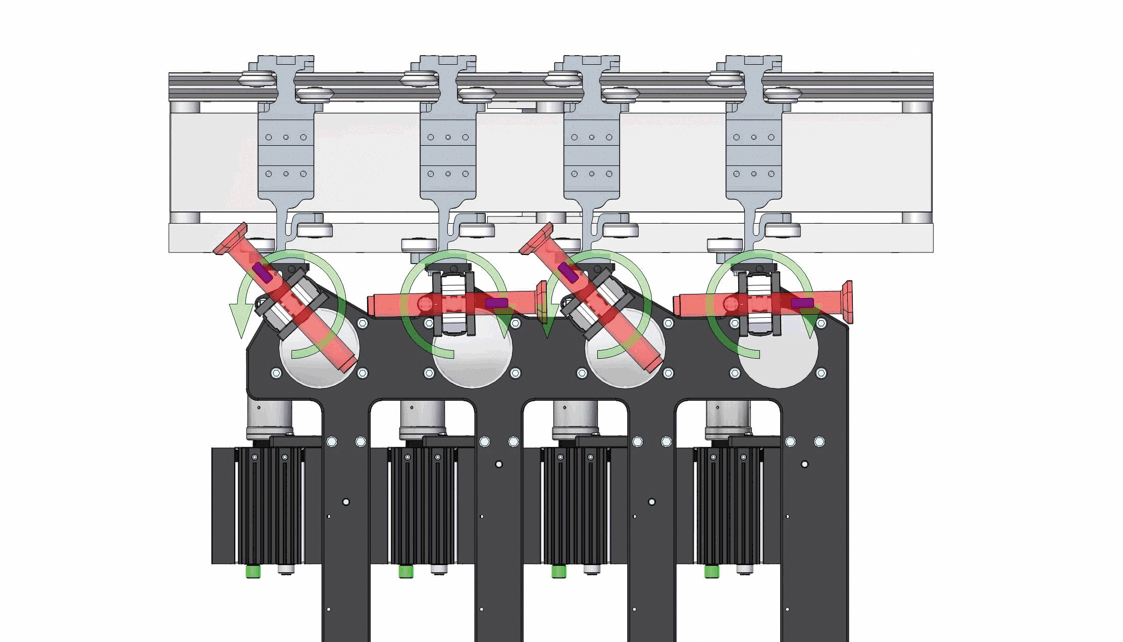
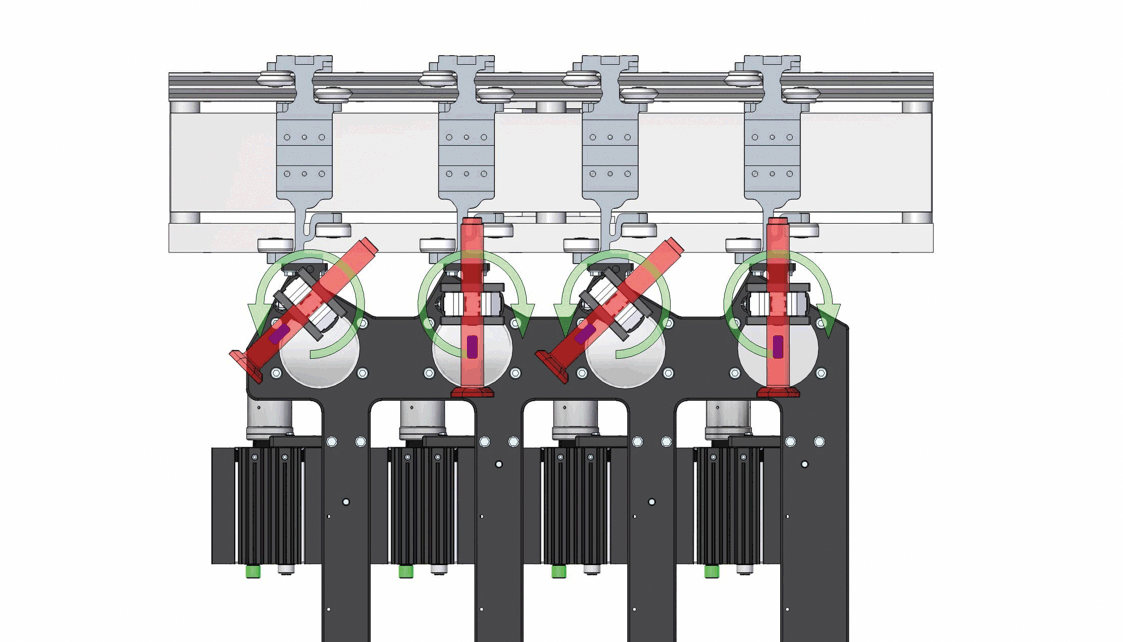
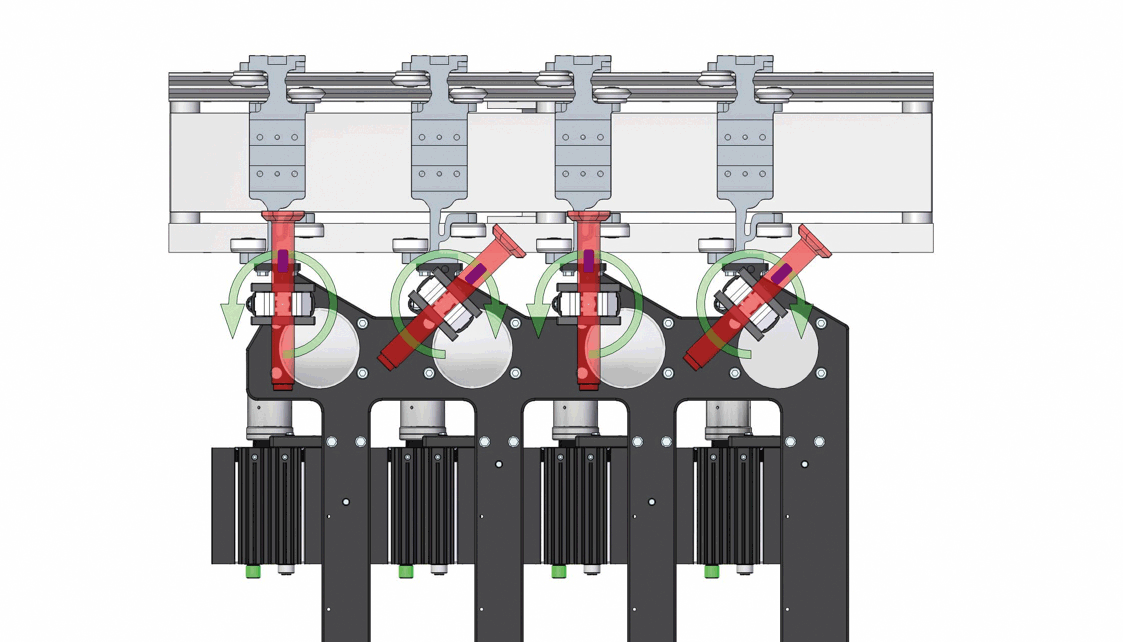
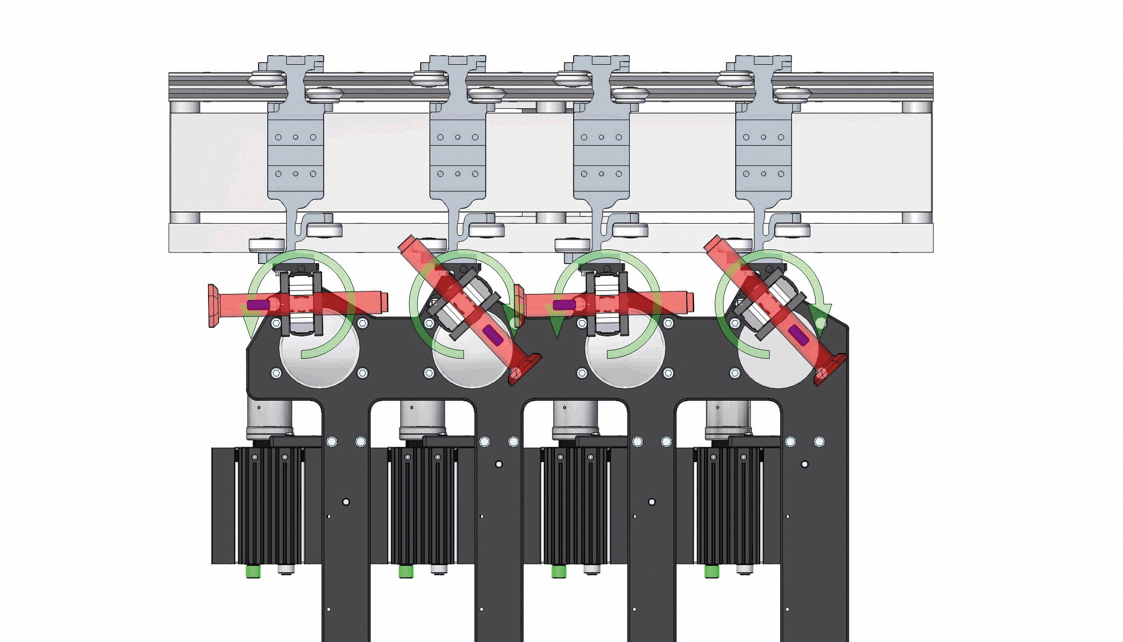
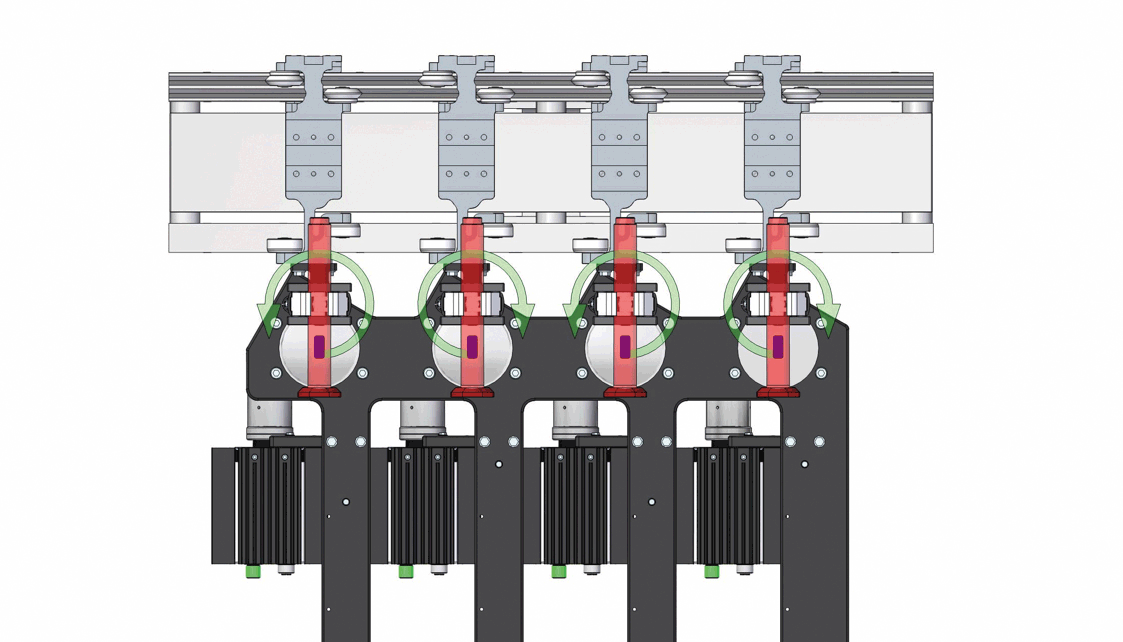
Demonstration of the inversion process for 4 out of 12 autoinjectors, with strategic orientation adjustments to prevent collisions. Image acquisition is performed statically after the upside-down inversion.
5. Tracking and Identification
5.1 Tracking trajectories of individual objects to identify swimming and sinking particles
This method's cornerstone is the detailed tracking of individual objects’ trajectories inside the liquid solution of the autoinjector:
- Image acquisition with the autoinjector stationary and the liquid still in post-inversion motion.
- More than 300 images acquired per autoinjector over 1 second.
- Trajectory tracking of all moving objects across the images.






Series of 6 images out of the full series of images acquired on a single autoinjector. The trajectories highlighted with a red line indicate objects moving downwards in the liquid solution, identified as foreign particles. The trajectory highlighted with a blue line indicates an object moving upwards, identified as a bubble. The two numbers in the fourth images from the left indicate the coordinate of a moving object.
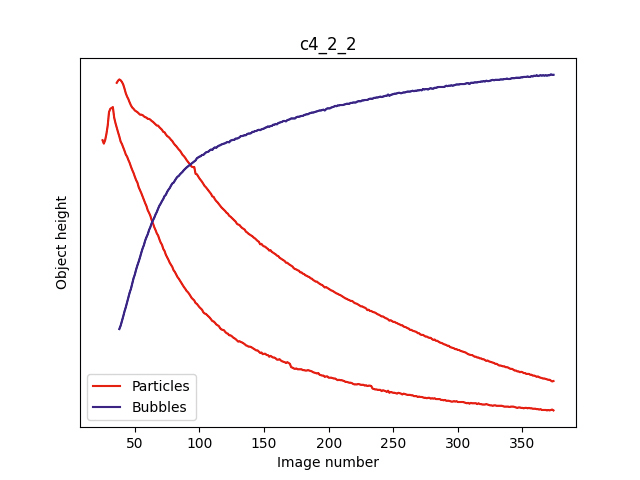
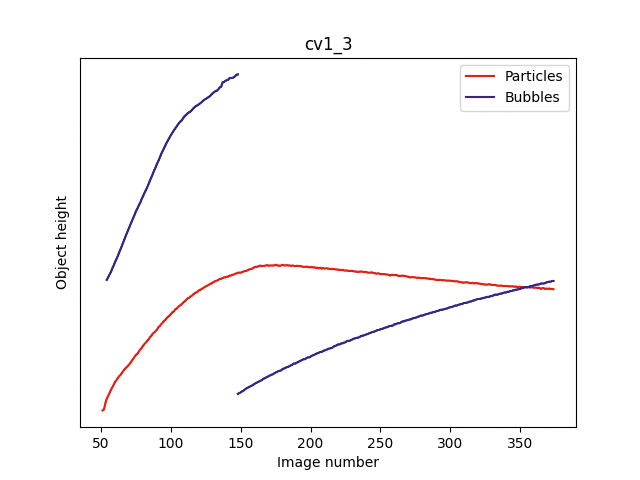
Vertical positions of objects in the acquired images as a function of the number of images taken. Two examples of two different autoinjectors are shown (left and right). In the first images of the series, objects tend to move all over inside the liquid solution, as an effect of the upside-down turning. In the last images, the objects moving downwards are identified as foreign particles (red trajectories), whereas the objects moving upwards are ascribed to bubbles (blue trajectories).
5.2 Identification of Floating Particles at the Fill Level
The behavior of foreign particulates inside a liquid solution can depend on the material they are constituted of and on the physical properties of the liquid solution itself. To identify particles floating at the meniscus a specific check has been added to the algorithm for the inspection of the fill level. This consists in the approximation of the meniscus profile with a certain number of lines and circle arcs, which can be tuned as a parameter. The smoothness of the profile can be directly correlated with the presence of particles floating at the meniscus. Additionally, overfill and underfill, as well as absence of liquid solution, are inspected in this test system, with two adjustable thresholds to take into account a gray zone and a reject zone.


Fill level profile represented through lines and arcs, showing the presence of a floating particle.


Fill level inspection illustrating thresholds for gray (dotted line) and reject zones (plain line).
6. Pioneering Complete AVI of a Fully Assembled Autoinjector
In addition to foreign particle detection, the system evaluates cosmetic defects across various autoinjector components, achieving the first comprehensive AVI of a fully assembled unit to our knowledge so far.
PRE-FILLED SYRINGE
- Sinking and swimming particles
- Floating particles and fill level
BASE AND CAP
- Inspection of all the parts in multiple test systems
BUTTON AND MECHANICAL PARTS
- Single mechanical pieces of various materials and colors checked for cosmetic defects
- Check of button color

7. Conclusion
WILCO AG's methodology successfully identifies foreign particulate matter inside autoinjectors by tracking single objects' trajectories. This approach significantly reduces the risk of false rejects due to harmless gas bubbles, achieves additional time for image acquisition and analysis without compromising machine speed, and is ready for implementation in platforms combining AVI with leak detection for difficult to inspect parenterals (DIP). Moreover, its compatibility with deep learning for image analysis promises to enhance detection rates and reduce false rejects even further.
Authors:
Dr. Chiara Sinito, Dr. Oliver Richter, Markus Heller, Daniel Sommer, Dr. Matthias Kahl - WILCO AG
References:
Pictures: WILCO AG