Pharma Lab Services for reliable Inspection
Our engineers are ready for your challenges. From feasibility and method development studies to system design for visual inspection or our own CCI studies to solutions for HSA calibration standards or positive and negative controls. We make sure that you have the best solution to achieve the greatest possible process security.
Feasibility Studies
Would you like to introduce a new packaging material or product? Then an early clarification of the possibilities of a leak test or visual inspection is central. We are happy to support you in this process and carry out the corresponding feasibility studies for you.
Maybe you already have a concept for leak testing and would now like to check its feasibility. Do the planned changes require an adaptation of the existing inspection strategy or is a new test procedure necessary? We support you in answering these questions. We can also evaluate the maximum possible performance in the context of a feasibility study in advance of a project.
A wide range of possibilities

These feasibility studies range from a non-binding "try it out" to a detailed study including a proven strategy for the generation of positive controls and their use in connection with the proposed technology.
From non-binding "try out" with the result “seems feasible” to detailed studies and test setups, we will find the right set-up for your project.
Method Development
The task of implementing a validated leak test strategy is not an easy one. There are many influencing factors on the measurement technique, including the pre-treatment of the packaging material, the environmental conditions and the physical properties of the filler, to name just a few.
These factors are central to a reliable CCI strategy, which is why an overall concept based on a solid knowledge base should be developed in advance.
Method development as a helpful tool

At the beginning of the process, all relevant steps are defined and the required information is gathered. The results of this process analysis are then mapped in a PFMA. Based on the risk analysis, the control strategy is defined and the appropriate test procedures are selected. Thanks to our many years of experience, we can develop the optimal method for your specific process so that it can be smoothly validated and implemented later.
In order to implement a reliable CCI strategy all these points have to be considered. Therefore, if possible, an overall concept based on a solid knowledge base must be created in advance of the choice of a solution.
Visual Inspection System Design
For the visual inspection of parenteralia in various packaging materials, we rely on an individual system design in six phases. The customer-specific setup is developed in an intensive preparation phase and then incorporated into the machine design. This enables us to achieve the greatest possible reliability of the inspection results.
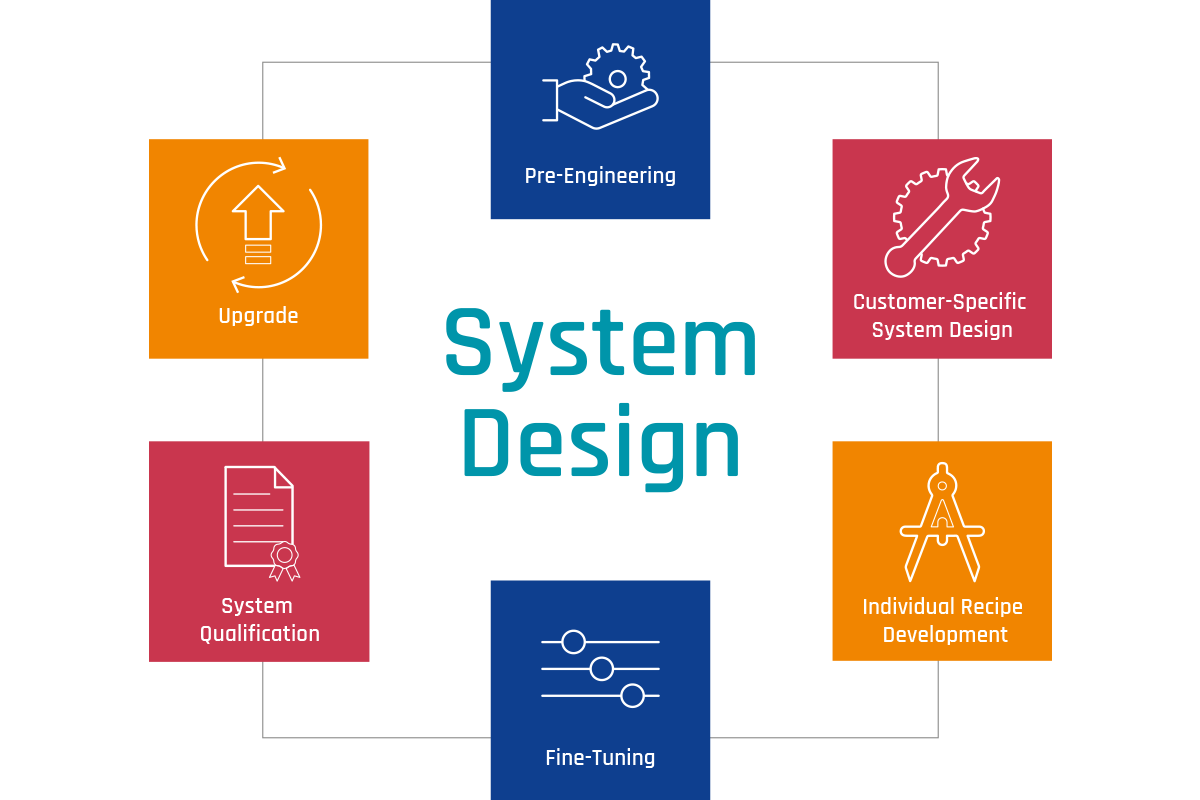
Pre-engineering as the key to success
A well-founded analysis of your needs creates the basis for an individual solution for visual inspection. For this purpose, a manual inspection is carried out in our own laboratory based on your test criteria and the corresponding test sets, consisting of good and bad samples. The measurement results are then verified and documented. They form a common understanding of the defined inspection tasks.
The holistic analysis also includes further test method procedures for leak testing, Headspace Analysis (HSA) or Near Infrared Spectroscopy (NIR). A combination of various inspection technologies with camera and X-ray-based visual inspection can significantly increase process reliability.
Customer-specific system design
Based on the knowledge gained from pre-engineering, the required hardware components are defined and a customer-specific optical design is created. We exclusively use state-of-the-art components. This is followed by the recording of image sequences in the laboratory, which are collected in a corresponding database. This database is used to evaluate the most powerful algorithms for inspection at each individual test station. A high degree of individualization is possible for both the software and the hardware. You have a large selection of algorithms, cameras and lighting at your disposal which can be individually combined for your project.
Individual recipe development
In a further process step, the image data is evaluated in detail. The individual parameters are optimized with the aid of a large number of preconfigured tools, thus increasing the inspection accuracy and performance (false reject rate and detection rate). For this purpose, specifically developed deep learning algorithms are used, for example to improve the differentiation between air bubbles and particles. The result is a customer-specific recipe.
In this phase, the optical components such as camera position, light setting, hardware, etc. are also defined. The optical path always remains the same. In this way, there are no moving parts such as mirrors or cameras, which reduces the susceptibility to errors to a minimum and increases the service life of the components.
In addition, the optical path is developed in such a way that the entire format range is always covered. This eliminates the need for further adjustments to the optical design, eliminating another potential source of error.
Fine tuning
Based on the test results and your requirements, we now define the automated visual inspection solution. The recommendations of the optical design and the software algorithms are documented for you. With the help of the integrated offline mode, a fine tuning of various form parameters is carried out and new form parameters are generated. Due to the flexible alignment and modular design, transfer from the laboratory to the production system is easily possible.
Qualification of the system
Our vision team will support you during the qualification of your system. Function and qualification tests are carried out and support for the Knapp test is also part of our range of services. A comprehensive training for your team concludes the qualification.
Longterm sustainable use assured
Due to existing inspection functions and the modular system design, upgrading to new formats is extremely easy. Our vision team can help you implement new inspection functions or expand to new formats and products.
Inherent CCI Studies (HE-Leak)
The sterility of an ingredient can be guaranteed by the tightness of pharmaceutical packaging materials.
The development of a suitable packaging material for a drug and thus the selection of the appropriate components are tasks that should be addressed as early as possible in the commercialization process. One of the requirements is that the different components ensure the tightness of the packaging material.
In inherent CCI studies, leaks are determined using helium mass spectroscopy or helium leak detection. This technology has the highest detection limit and detects even the lowest leak rates, which is a clear advantage. In addition, helium mass spectroscopy makes it possible to identify leakage rates that correspond to the MALL (maximum allowed leakage level), as described, for example, in the publication of L. Kirsch and referred to in the USP1207.
HSA Calibration Standards
Calibration standards are important for the validation of your devices.

Our HSA calibration services give you the confidence that every vial has been measured and remains within the specified tolerance ranges. Our qualified team of experts will confirm that the reference vial can be used to calibrate your machines for use in GMP environments.
We recommend that vials are recalibrated at regular intervals.
Positive / Negative Controls
Positive or negative samples form the basis for flawless and accurate measurement results.
We create test samples with controlled microleaks for positive control of your test system. If your machine detects these test samples as faulty, you can be sure that your machine is functioning properly.
We will be pleased to advise you on the different types of positive controls to be selected depending on the application and product.
Engineering Services
As a specialist for special and small series machines, we have our own development departments for design, software and pneumatics. This enables us to offer everything from a single source throughout the entire process and to react quickly to your individual requirements. This is why we can offer you flexible and taylor-made solutions.

For the production of specific parts, we work together with strategically selected partners. Assembly and commissioning are in turn carried out entirely by us. The entire system is set up and put into operation and the perfect functioning of mechanics, electronics, software and measurement technology is ensured. For efficient use, we use the standardization and modularization of proven subsystems in combination with new developments. New technologies, as well as product or customer specific requirements, are always the drivers for new developments.
Our engineering services form the basis for your individual solutions.
Need more details about our 360° service offering?
We are happy to advice you personally.