Determination of Water Activity with Near Infrared Spectrocopy
Abstract
Water activity is a critical quality attribute influencing the chemical stability, microbial safety, and shelf life of pharmaceutical products. The concept of water activity first emerged in the food industry in the late 1960s and has since been extensively studied. The pharmaceutical industry incorporated it into its regulatory framework through its inclusion in the European Pharmacopeia Ph. Eur. and United States Pharmacopeia (USP). It is a fundamental indicator of product stability and safety. As such both USP <922> and Ph. Eur. 2.9.39 mandate water activity testing from early formulation and packaging development to manufacturing, quality control, and long-term stability testing. Monitoring water activity during development and production of pharmaceuticals enables risk-based control of moisture-driven instability and microbial growth. Therefore, USP as well as Ph. Eur. place strong emphasis on the use of validated analytical instruments for water activity determination in modern pharmaceutical manufacturing.
This study presents a comparison of two non-destructive technologies recognized in USP <922> as suitable methods for determining water activity, namely Near Infrared (NIR) spectroscopy and Headspace Analysis (HSA). While HSA enables direct determination of water activity through water vapor pressure measurement, NIR offers a fast, non-destructive, and multi-parameter alternative capable of simultaneously assessing water activity, moisture content, and material composition. The objective of this work is to demonstrate the suitability of NIR spectroscopy as a robust and regulatory-compliant technology for 100% in-line water activity screening in pharmaceutical products. This is achieved by benchmarking NIR-predicted water activity values against established HSA reference measurements on the same samples.
Download White Paper
1. Introduction
Water activity (aw) represents the ratio of the equilibrium vapor pressure of water in a product headspace (P) to the vapor pressure of pure water (P₀) at the same temperature (T) [1]:
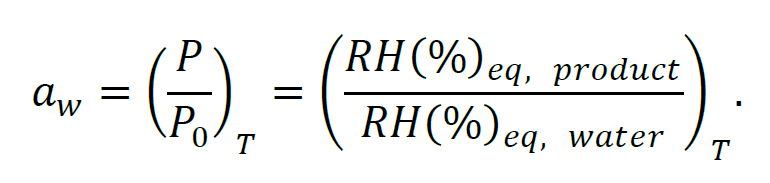
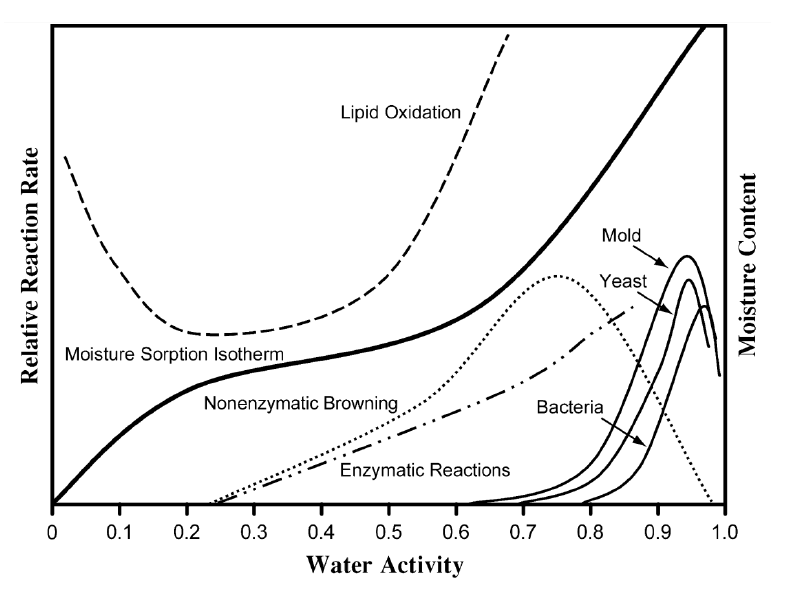
Where RH(%)eq is the headspace equilibrium relative humidity expressed in percentage above the product or above pure water in a sealed container. As the moisture content of a sealed sample is closely linked to the water vapor pressure in its headspace, and consequently to water activity, NIR can indirectly determine water activity by measuring the sample moisture content. This relationship between moisture content and water activity is described by sorption isotherm, Fig. 1. Thus, sorption isotherm builds the foundation for linking moisture content information (measured e.g. by NIR) with water activity values (measured e.g. by HSA) [2].
2. Methods
Near Infrared (NIR) Spectroscopy
NIR relies on molecular overtone and combination vibrations, of primarily O–H bonds, whose absorbance intensities correlate with the moisture content of the sample. Based on the NIR spectra, partial least squares (PLS) regression models are developed to quantify moisture content and, consequently, to determine water activity.
Tunable Diode Laser Absorption Spectroscopy (Headspace Analyzer - HSA)
Laser based headspace analysis (HSA) is a USP<922> compliant method to measure water activity directly in the headspace of a product. It quantifies the water activity by measuring and comparing the equilibrium water vapor pressure in the product headspace to that of pure water at the same temperature. Instruments are calibrated with saturated salt solutions as specified in regulatory guidelines to ensure traceable and accurate readings.
3. Results
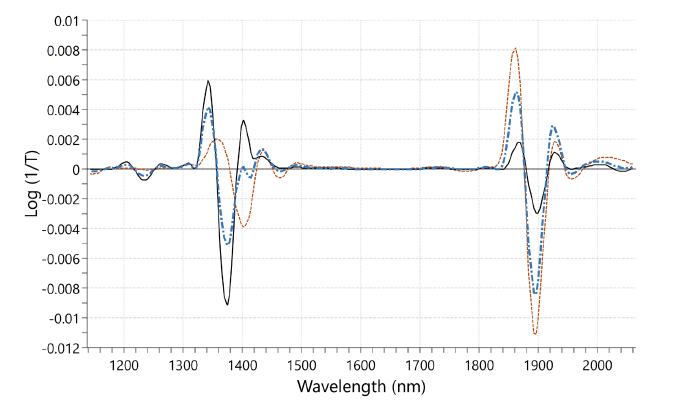
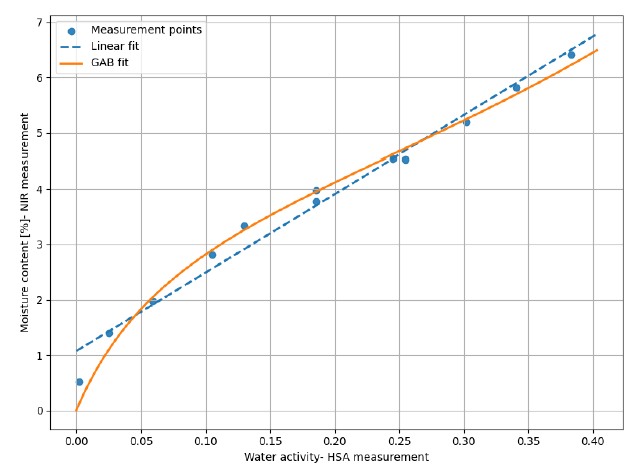
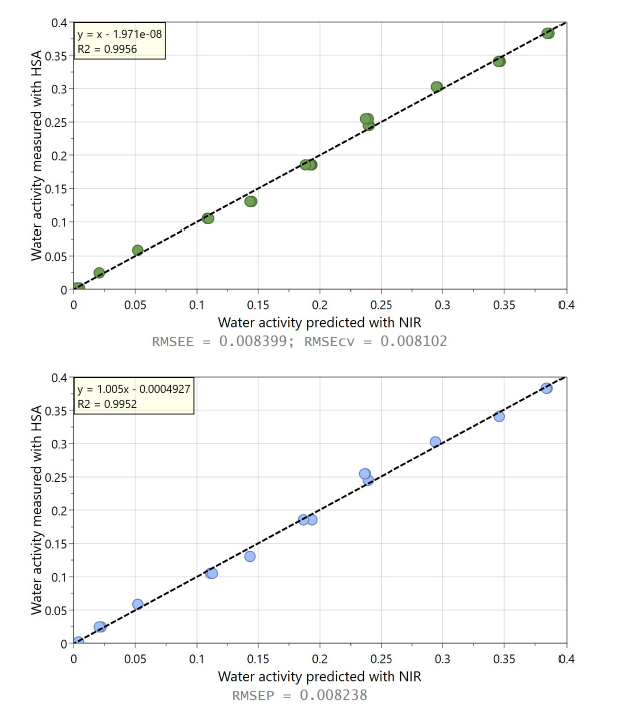
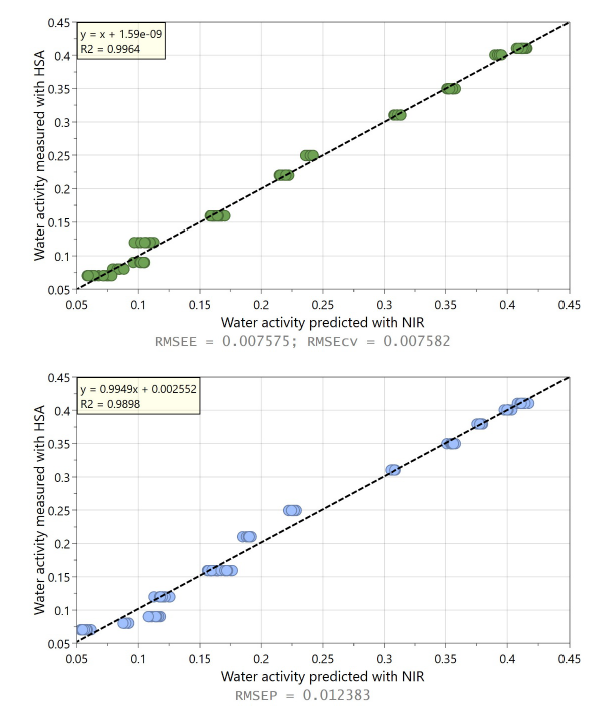
In this comparative study silica gel (SiO₂; Sigma-Aldrich, high-purity grade, 60 A, 35-60 mesh) and a lyophilized pharmaceutical product were used to assess the accuracy of NIR for water activity measurement. NIR spectra are obtained using in-line NIR spectrometer, SPECTRA NIV; WILCO AG; Wohlen, in diffuse-reflectance geometry. Fig. 2 shows second derivative NIR spectra for SiO₂ with three different moisture contents, where variation in O-H bond formation leads to intensity changes in certain spectral bands. These spectral features were captured by a PLS regression model calibrated with Karl Fischer titration reference method. These PLS models were eventually used to noninvasively predict moisture content of new samples not previously seen by the model. For the same SiO₂ samples, water activity was measured in parallel using HSA laboratory instrument, NEO HSX; WILCO AG; Wohlen. The HSA derived water activity values are compared with the moisture content data obtained from the NIR spectrometer in Fig. 3. It should be noted that the extent and type of such correlation – linear or non-linear- depend on the product and can be comprehensively interpreted by the sorption isotherm of that specific substance.
A strong correlation was observed between the HSA obtained water activity and NIR obtained moisture content values for SiO₂ (Fig. 3) and for the Lyophilizate product (data not shown). This correlation served as the basis for developing a PLS regression model to predict water activity from NIR spectra.
Figures 4 and 5 demonstrate the prediction performance of the NIR based PLS regression model, for SiO₂ and Lyophilizate product respectively. Highly precise predictions from the in-line NIR spectra based PLS regression models were obtained with an inaccuracy in water activity determination of below 0.009 for SiO₂ and 0.01 for the lyophilized product. Building on previous studies mainly conducted under laboratory conditions [4–7], these results demonstrate strong NIR spectroscopy performance for in-line water activity determination.
4. Conclusion
According to the USP <922> and Ph. Eur. 2.9.39, water activity is an essential parameter for ensuring the stability and quality of pharmaceutical products. Supported by literature, USP <922> and the experimental evidence presented in this work, NIR-spectra based PLS regression models can precisely predict water activity values in pharmaceutical and non-pharmaceutical products. This capability of NIR to precisely determine water activity, in addition to the moisture content and chemical composition, from a single noninvasive spectral measurement demonstrates its strong analytical potential. It supports the adoption of NIR as primary regulatory-compliant analytical technology for 100 % in-line screening in pharmaceutical manufacturing environments.
Authors:
Peyman Zirak, Inna Gerber, David Stäger - WILCO AG
References
[1] United States Pharmacopeia. USP <922> Water Activity. In United States Pharmacopeia and National Formulary (USP-NF); United States Pharmacopeia convention: Rockville, MD, 2020
[2] Iji, S. I., et al. (2025). Models for the Development of Sorption Isotherms: A Review. American Journal of Food Science and Technology, 13(2), 27–37.
[3] Labuza, T. P. et. al, (1970), Water content and stability of low moisture and intermediate moisture foods, Journal of food technology, 24, 543-550.
[4] Rongtong, B., et al. (2018). Determination of water activity, total soluble solids and moisture... Agriculture and Natural Resources, 52, 557–564.
[5] Christy, A. A. (2010). New insights into the surface functionalities and adsorption evolution of water molecules on silica gel surface. Vibrational Spectroscopy, 54, 42–49.
[6] Nielsen, O. F., et al. (2012). Water Activity. Spectroscopy: An International Journal, 27(5–6), 565–569.
[7] Achata, E., et al. (2015). Near Infrared Hyperspectral Imaging for Monitoring Moisture Content and Water Activity in Low Moisture Systems. Molecules, 20, 2611–2621.
[8] Anderson, R. B. (1946), Modification of the Brunner, Emmett and Teller equation, Journal of the American Chemical Society, (68), 686-691.
Pictures: WILCO AG